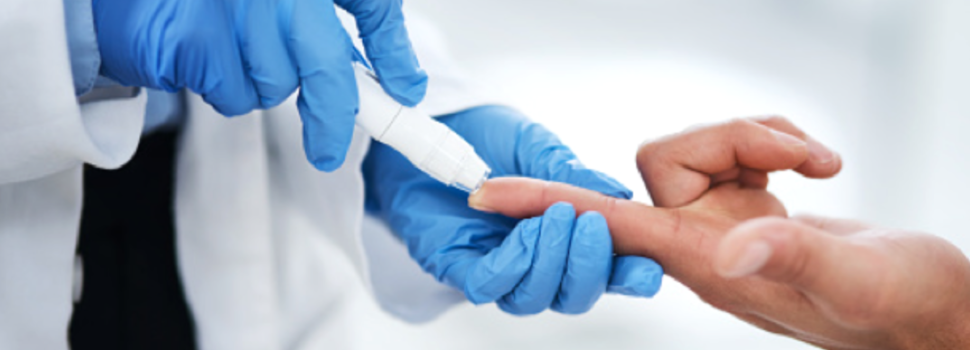
A century after Frederick Banting and Charles Best’s insulin discovery in the U of T lab, Toronto’s Sunnybrook researchers are working on a new approach to cure Type 1 diabetes.
Insulin discovery in 1921 turned diabetes from a deadly disease into a manageable one for millions of people worldwide. Even though medical and technological advances have come a long way since then, integrating glucose monitors, insulin pumps and smartphone detectors in our lives, both Dr. Rahul Jain and Dr. Robert Screaton say diabetes is still a health crisis for many Canadians.

According to the 2019 Diabetes Canada Cost Mode data, one in three Canadians has diabetes or is pre-diabetic. Adults of 20 years and older have a 50 per cent chance of acquiring diabetes, while under 50 per cent of Canadians can identify less than half of the early signs of diabetes. This means more than half of the Canadian population is unaware that symptoms such as hunger, fatigue, thirst, frequent urination, blurry vision, dry mouth and itchy skin can be early signs of diabetes.
More than 90 per cent of diabetes cases are Type 2 diabetes (T2D), usually seen in adults and recently increasingly seen in children. According to Diabetes Canada, its alarming high prevalence calls for urgent action and raising awareness about its symptoms and causes.
Dr. Jain, an academic Family Physician and Hospitalist at Sunnybrook Health Sciences Centre and Assistant Professor in the Department of Family and Community Medicine at the U of T, says T2D can be managed with health behaviour changes, and occasionally oral medications and/or injections, including insulin. However, he says it is important to start screening people for diabetes at the age of 40, even earlier, if they have risk factors. These factors include certain pre-existing health conditions or patients coming from Indigenous or South Asian populations, says Dr. Jain. He says timely diagnosis can prevent developing the disease before substantial damage is done to the body.
“Some people who have very high sugar levels in the blood may develop symptoms like fatigue, blurry vision, increased thirst and frequent urination. However, this is often when the sugars are very, very high. So it’s important to recognize that not all people with diabetes will present with symptoms. And that’s why it’s important to screen. When we think about it, the whole purpose of treating diabetes is to prevent complications, especially on micro and macrovascular systems,” says Dr. Jain, “this can put patients with diabetes under the risk of developing problems of the eyes, kidneys, nerves, even heart attack or a stroke.”
Type 1 diabetes (T1D), however, makes up about 5 -10 per cent of all people having diabetes. T1D is an autoimmune disease, with genetic and environmental triggers, and is out of the patient’s control, says Dr. Robert Screaton, senior scientist in Biological Sciences at Sunnybrook Research Institute and an Associate Professor in the Department of Biochemistry at the U of T. He says the problem with T1D is that patient’s immune system starts to attack the beta cells of the pancreas that make and secrete insulin after a meal. Once the number of beta cells suddenly drops to a dangerously low level, not enough remains to secrete the necessary amount of insulin to maintain blood sugar at normal levels, causing blood sugar levels to go up. Once these cells are lost, they do not grow back. This is why patients with Type 1 diabetes need regular insulin injections to survive.
Sunnybrook researchers are now pioneering a new approach to treat T1D. The research is aimed at figuring out why lost insulin-generating beta cells cannot regenerate. Dr. Screaton says a whole series of genes that sit at the cell surface and are involved in cell-to-cell and tissue-to-cell communication were removed one by one from human beta cells to see if they could start to proliferate. “Using robots and genetic techniques, we found a series of cell surface receptors that, when removed, resulted in beta cells starting to replicate,” says Dr. Screaton.
The next steps would be using chemical biology or a pharmacology approach to see if it is possible to find an inhibitor of these genes, add it to cells and see the same thing.
“If we can do that, then we can start testing this in animals, and then eventually in humans,” says Dr. Screaton. However, he adds there is a long road ahead, and if the research is successful, the hope is to reduce the standard 20 or 30 year time period it takes to develop, approve, and get the drug out in the market.
